

Overview
There are a few ways in which you can count cells to prepare them for passaging or other experiments. Though automated cell counters save time when quantifying cell numbers, hemocytometers are a budget-friendly way to perform this same task. In this protocol, you will learn how to count cells with a hemocytometer.
Materials
- Cell Pellet
- Cell Medium
- Trypan Blue (optional)
- Hemocytometer
- Cell Counter
- Cover Slips
- Microscope
- Micropipettes
- Micropipette Tips
Methods
- Warm culture medium to 37˚C;
- After preparing a cell pellet (refer to Preparing a Cell Pellet), resuspend it in 1-5 mL of culture medium, making note of the media volume added;
- If you wish to also quantify viability, follow this protocol to learn how to mix your cells with trypan blue;
- Pipette 10 µL of your cell suspension and place into one hemocytometer chamber;
- Carefully place a coverslip on top, ensuring that no bubbles are trapped in the system;
- Place your hemocytometer under a microscope;
- Using the cell counter, count the cells in the squares outlined below. Make sure to NOT count cells that are sitting on dotted lines. If you used trypan blue, make sure to differentiate counting for live (normal) and dead (blue) cells;
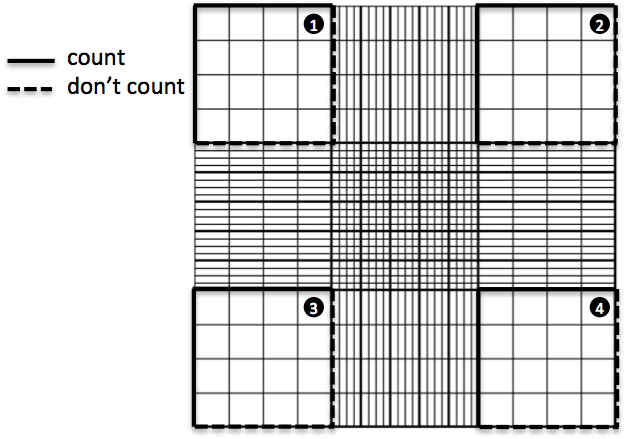
- Use the following formula in order to calculate the number of cells you have in your suspension: (total cells counted)/(4 squares counted)*10-4*initial volume*dilution factor = total number of cells;
- Note: 10-4 is the volume of squares on the hemocytometer (0.1 mm3). The dilution factor will be 1, unless you have diluted your cell suspension with trypan blue.
- You may also use this hemocytometer calculator.
- Now that you have your cell number, you may follow this protocol to prepare a cell pellet for bioink encapsulation or other protocols, depending on your application.

