
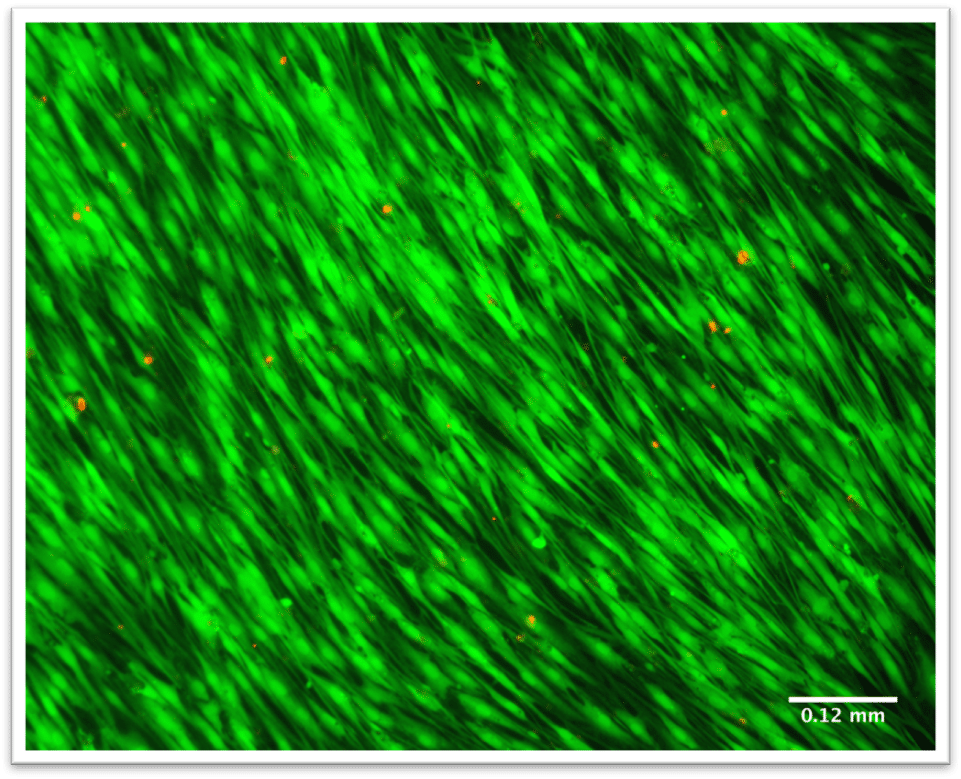
Overview
Skin engineering is a prominent alternative to traditional methods used for wound healing, drug testing & discovery, and other skin-related applications. However, challenges remain when it comes to generating complex skin substitutes. Follow this step-by-step protocol for bioprinting fibroblasts.
Collagen is extremely difficult to pattern in optimal concentrations (3 mg/mL). With Allevi’s exclusive CORE™ printhead and this skin bioprinting protocol, you are now able to print and pattern pure 3 mg/mL type I collagen or 8 mg/mL methacrylated collagen. This is the first time that such low concentration collagen can be printed and patterned through 3D bioprinting. Allevi’s skin bioprinting protocol presents a novel solution to automating and standardizing the creation of skin models.
Materials
- Allevi printing dish
- 10 mL of Advanced Biomatrix PureCol
- Allevi 5 mL syringe
- 2 x Allevi layering tips™
- Syringe coupler
- NaOH
- 10X PBS
- Human neonatal dermal fibroblasts or human adult dermal fibroblasts
- Note: you may also add keratinocytes and melanocytes
- Fibroblast growth medium
Methods
- Follow this protocol to prepare PureCol® and your cells for printing;
- Set your Allevi extruder to 4˚C;
- Attach the Allevi layering tip™ to your syringe;
- Bioprint on the Allevi printing dish;
- If you have an A3, set your bed plate to 37˚C to crosslink your skin layers at the time of deposition;
- Incubate your structure at 37˚C for at least 30 minutes;
- Culture your layered tissue as desired.
Print Settings
| Speed (mm/s) | Layer height (mm) | Nozzle Diam (mm) | Gauge |
| 5 | 0.2 | 0.2 | 27 |
| Pressure (PSI) | Crosslink (sec) | Print Temp (°C) |
| 10 | – | 4 |
We hope that you found this step-by-step protocol for skin bioprinting useful! You are now ready to image and analyze your 3D bioprinted skin construct – click here for a full list of analysis protocols.
References
Lee, V., Singh, G., Trasatti, J. P., Bjornsson, C., Xu, X., Tran, T. N., … Karande, P. (2014). Design and Fabrication of Human Skin by Three-Dimensional Bioprinting. Tissue Engineering Part C: Methods, 20(6), 473–484. https://doi.org/10.1089/ten.tec.2013.0335

