
Introduction
Poly(lactic-co-glycolic acid), or PLGA, is a common thermoplastic used in bioprinting for its tunable degradable and mechanical properties[1,2]. The degradation and mechanical properties of this material can be altered by adjusting the ratios of the copolymers polylactide and polyglycolide [1,2].
Thermoplastics in bioprinting are often used as a support structure to provide extra mechanical strength and as reinforcement for matrix bioinks[1,2]. Polycaprolactone is a biodegradable thermoplastic used in a variety of medical applications, including bioprinting. This study tested the viability and print resolution of PLGA with the Allevi 2.
See Also
- Bioprinted Alginate Viability
- Bioprinted Collagen Viability
- Bioprinted GelMA and LAP Viability
- Bioprinted PEGDA Viability
- LAP and Irgacure Viability with GelMA
Results
Viability Data
ATP of thin films demonstrates an increase in ATP from day 1 to day 3, with statistically higher ATP values on day 3 and day 7 when compared to day 1. Likewise, LIVE/DEAD images confirm viable cultures of HNDFs seeded on PLGA thin films after 1,3 and 7 days of culture.
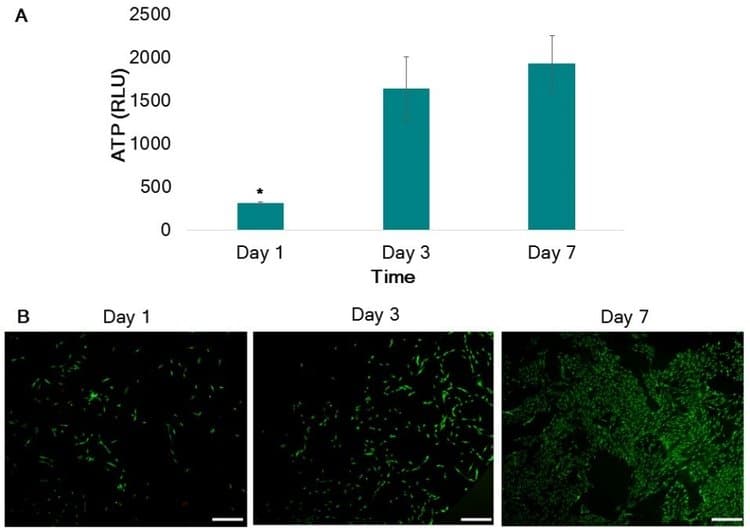
These results demonstrate the ability of PLGA to support viable cultures and create consistent, high-resolution 3D geometries. This support material can be used in combination with matrix bioinks or on its own to create 3D culture environments for tissue engineering.
Print Resolution


PLGA was printed using print parameters and settings provided by Allevi (Figure 2). Line width average was 0.15 ± 0.01 mm.
Methods: Bioprinted PLGA
Cell Culture
Primary Human Neonatal Dermal Fibroblasts (HNDFs) from ATCC were cultured at 37 °C and 5% of CO2. HNDFs were cultured using Dulbecco’s Modified Eagle Medium (Corning) supplemented with 10% fetal bovine serum (Hyclone) and 1% penicillin-streptomycin-amphotericin (Corning). Passage numbers under 10 were used.
Thin Film Fabrication
Viability of PLGA was tested with cell seeding of human neonatal dermal fibroblasts. First, PLGA was sterilized with UV light overnight. Then, thin films were fabricated by melting PLGA at 100°C on a glass coverslip, then flattening with a second glass coverslip. Thin films were then allowed to cool to room temperature before removal from coverslips.
HNDFs were suspended at a concentration of 9500 cells mL-1 and pipetted into the wells containing the PLGA thin films. Enough cell solution was used to submerge the samples. After overnight incubation, media was exchanged.
Viability Assessment
To quantitatively assess cell viability, CellTiter-Glo 3D ATP Assay (Promega) was performed on days 1,3 and 7 of culture according to the manufacturer’s protocol and analyzed using a BioTek Synergy 2 Plate Reader. A LIVE/DEAD kit (Life Technologies) was used to qualitatively assess the viability of samples.
Statistical analysis was performed using an Analysis of Variance (ANOVA) test to determine if differences were present amongst treatment groups. If differences were determined from the ANOVA, a post hoc Tukey’s multiple comparison test was used to determine statistical differences between groups tested. A confidence level of 95% (α=0.05) was used for all analyses. Error bars on graphs represent the standard deviation from the mean.
Bioprinting
PLGA was bioprinted using print parameters and settings provided by Allevi. Line width and pore size were analyzed with ImageJ software. Lattice and line designs were each printed 3 separate times. 3 images of each print were taken, then three measurements were taken from each image.
References
- [1]Pati, Falguni et al, “Ornamenting 3D Printed Scaffolds with Cell-laid Extracellular Matrix for Bone Tissue Regeneration” Biomaterials, 2015(37), pp. 230 – 241.
- [2]Castro, Nathan J. et al, “Integrating biologically inspired nanomaterials and table-top stereolithography for 3D printedbiomimetic osteochondral scaffolds” Nanoscale, 2015 (7), pp. 14010-14022.

